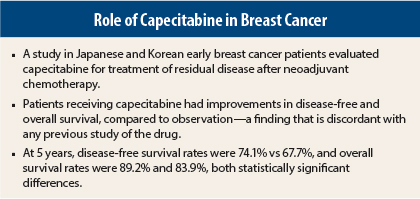
Treatment with capecitabine increased disease-free and overall survival in breast cancer patients with residual disease after neoadjuvant chemotherapy, according to a study reported by researchers from Japan and Korea at the 2015 San Antonio Breast Cancer Symposium.1
“It has been unclear whether postoperative systemic chemotherapy following neoadjuvant chemotherapy is able to prolong survival. These first efficacy results show that survival is significantly improved by the addition of capecitabine to standard therapy,” said Masakazu Toi, MD, PhD, Professor at Kyoto University Hospital in Japan. Dr. Toi is founder and Senior Director of the Japan Breast Cancer Research Group.
Clifford A. Hudis, MD, Chief of the Breast Medicine Service at Memorial Sloan Kettering Cancer Center, New York, and an ASCO Past-President, considered the investigation a highlight of the symposium’s presentations.
“The study showed an overall survival improvement. If this holds up, I think this is a very important observation,” Dr. Hudis told The ASCO Post.
He indicated that the results were discordant with previous studies of the drug—such as the German study presented here last year by von Minckwitz et al2—a point that was reinforced during the discussion by a number of other breast cancer experts who peppered Dr. Toi with questions.
Details of CREATE-X
The CREATE-X study, conducted at 84 sites, enrolled 910 patients with HER2-negative breast cancer and residual invasive disease (no patients with pathologic complete response or node-positive disease) after neoadjuvant treatment with an anthracycline and/or taxane. Most patients (80%) received an anthracycline followed by a taxane, and approximately 60% had prior fluorouracil (5-FU). Adjuvant endocrine therapy was received by approximately 40% of premenopausal patients and 25% of postmenopausal patients.
The 455 patients randomly assigned to active treatment were given capecitabine at 1,250 mg/m2 twice daily, 2 weeks on and 1 week off, for up to eight cycles. Six cycles were completed by 58%, and eight cycles were completed by 38%. Dose reductions were required by 24% and 37%, respectively, whereas 18% and 25%, respectively, discontinued the drug.
Disease-Free and Overall Survival Improved
“In 2015, the first preplanned interim analysis was carried out at the point of 2 years’ follow-up from the last patient enrollment. The data monitoring committee recommended that this study be discontinued according to the protocol,” Dr. Toi reported.
At 5 years, the disease-free survival rate (the primary endpoint) was 74.1% with capecitabine compared to 67.7% in the control arm, a statistically significant 30% reduction in risk (one-sided P = .00524). Overall survival rates were 89.2% and 83.9%, respectively, a statistically significant 40% reduction in risk (one-sided P < .01).
The benefit was observed across subgroups, with hazard ratios ranging from 0.54 to 0.84. The hormone receptor–negative subgroup experienced a 42% reduction in risk with capecitabine, Dr. Toi reported.
Hand-foot syndrome associated with capecitabine was observed in 72.3% of patients; grade 3 hand-foot syndrome occurred in 10.9%.
Neutropenia and diarrhea were also significantly higher in the capecitabine arm. Neutropenia ≥ grade 3 was seen in 6.6% with capecitabine and 1.6% with observation; diarrhea ≥ grade 3 occurred in 3% and 0.4%, respectively.
During the discussion, Hope S. Rugo, MD, Director of the Breast Oncology Clinical Trials Program at the University of California, San Francisco, noted that the toxicity seemed less than is observed among U.S. patients and suggested that there might be pharmacogenomic differences. Dr. Toi acknowledged that capecitabine seems to be better tolerated among Asians, concluding, “The balance of benefit and toxicity would favor the use of capecitabine in the postneoadjuvant situation, but prediction for the therapeutic benefit needs to be investigated further.”
Brisk Discussion
The findings elicited brisk discussion at the meeting, mostly pertaining to the discordance between these and previous findings. Dr. Hudis pointed out, “Capecitabine as a single agent has not been that impressive in earlier studies, and prior studies in the adjuvant setting have not been positive. That’s what’s odd about this, and also that a fairly large proportion of patients had received 5-FU before they got capecitabine.”
Dr. Toi had no clear explanation, aside from the possibility that the outcomes derive from “non–cross-resistance to treatment with anthracyclines and taxanes.”
Richard D. Gelber, PhD, Statistical Director of the International Breast Cancer Study Group and Professor of Biostatistics and Computational Biology at Dana-Farber Cancer Institute, Boston, had a different take on the findings. In an interview with The ASCO Post, Dr. Gelber explained: “People are confusing the other studies of capecitabine. In those [negative] studies, patients either received capecitabine with other chemotherapy or as a single agent. This study is a different setting, and it’s more like maintenance.”
Dr. Gelber said the study is akin to the IBCSG Trial 22 (in press), where patients completed treatment with standard adjuvant chemotherapy, then were randomly assigned to observation or a single year of cyclophosphamide/methotrexate, metronomically dosed. Results of that study in the triple-negative disease subset were positive, as were the results here with capecitabine, he said.
“Maintenance [cyclophosphamide/methotrexate] improved outcomes for triple-negative cases,” he said. “The current trial is similar in the sense that they gave neoadjuvant chemotherapy, and patients not achieving a pathologic complete response continued on single-agent capecitabine, as the maintenance component.”
Steven E. Vogl, MD, a medical oncologist in the Bronx, New York, suggested that the study involved two different populations with different responses to the treatment.
“You have patients with estrogen receptor–positive disease, very few of whom achieve pathologic complete response and those actually don’t do badly,” he said. “Then you have a minority of patients, where a significant advantage of capecitabine is seen. Since the curves separate very early, and early relapse is a characteristic of triple-negative disease, I suspect most of your differences are driven by triple-negative disease. This may be telling us that for patients with triple-negative disease who don’t achieve a pathologic complete response after good neoadjuvant therapy, this [capecitabine] is probably a reasonable option,” he suggested.
He would not apply the findings to estrogen receptor–positive patients who lack a pathologic complete response. “I’d like to see these sets of patients separated out in the analysis,” he said.
In Dr. Hudis’ view, “The conservative thing to say is that we have gotten an exciting early look at these data, but like everything else, we need a final peer-reviewed manuscript to see the details.”
He concluded, “But make no mistake: Capecitabine is a broadly available, inexpensive agent, and this is an increasingly recognized setting—the lack of a pathologic complete response to preoperative treatment. Anything that improves overall survival has to be watched.” ■
Disclosure: Dr. Toi has received a research grant from Chugai Pharmaceutical Co. Drs. Hudis, Rugo, Gelber, and Vogl reported no potential conflicts of interest.
References
1. Lee S-J, Toi M, Lee ES, et al: A phase III trial of adjuvant capecitabine in breast cancer patients with HER2-negative pathologic residual invasive disease after neoadjuvant chemotherapy (CREATE-X, JBCRG-04). 2015 San Antonio Breast Cancer Symposium. Abstract S1-07. Presented December 9, 2015.
2. von Minckwitz G, Reimer T, Potenberg J, et al: The phase III ICE study: Adjuvant ibandronate with or without capecitabine in elderly patients with moderate or high risk early breast cancer. 2014 San Antonio Breast Cancer Symposium. Abstract S3-04. Presented December 11, 2014.