Case Summary presented by Steven M. Horwitz, MD, Assistant Attending, Lymphoma Service, Memorial Sloan-Kettering Cancer Center, New York.
This is a case of a 49-year-old woman who developed an enlarged right axillary lymph node. The woman had noticed some discomfort under her right arm, which she initially believed was an ingrown hair. Over a month or two, this grew to the size of a chickpea. The woman’s primary medical doctor ordered a mammogram, which was unremarkable in the breast but confirmed a 1.5-cm right axillary node enlargement. A CT scan of the chest showed the right axillary node as well as some subpectoral lymphadenopathy and a lung nodule. An excisional right axillary node biopsy was performed and showed ALK-negative anaplastic large cell lymphoma. At this point, the woman was referred to our center.
Workup
A PET/CT scan showed increased FDG uptake in the remaining subpectoral node as well as the lung nodule. Fine-needle aspiration of the lung nodule showed an anaplastic large cell lymphoma, with large CD30-positive T cells. A bone marrow biopsy was negative. Her lactate dehydrogenase (LDH) level was elevated. This was a young woman with stage IV disease, high LDH, and an International Prognostic Index of 2.
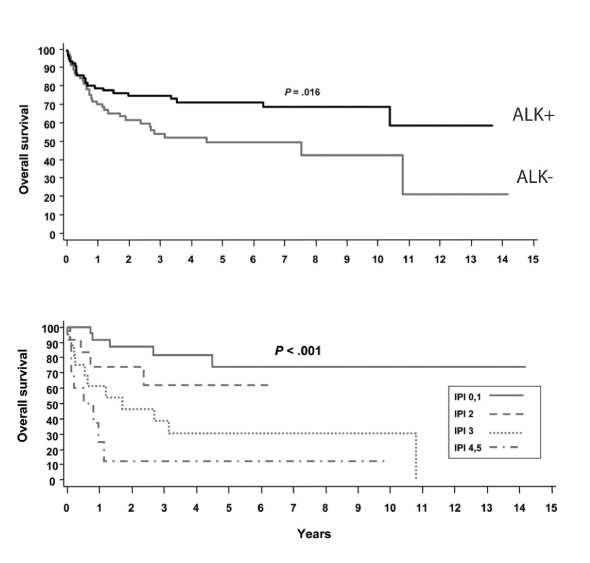
In general, we think of ALK-negative anaplastic large cell lymphoma as a fairly aggressive, poor-prognosis lymphoma that we generally treat more aggressively than ALK-positive anaplastic large cell lymphoma (Fig. 1). CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) is probably the most commonly used regimen in this setting. There is a sense that etoposide in addition to CHOP, or CHOEP, adds benefit. It was recommended that this patient receive CHOEP followed by an autologous stem cell transplant.
The woman was started on CHOEP and tolerated this reasonably well. She initially started on an every-2-week schedule, but it was extended to every 3 weeks due to hematologic toxicity. Repeat staging after four cycles showed a PET-negative complete response. Her stem cells were collected, she proceeded to high-dose therapy and autologous stem cell transplant, and she did well. However, routine imaging at 1 year showed new lymphadenopathy and biopsy-confirmed recurrent disease.
Discussion
At this point, patients with T-cell lymphoma or anaplastic large cell lymphoma are left with a little bit of a conundrum in the sense that we really do not have strong data to guide us. Two of the drugs approved in this setting, pralatrexate (Folotyn) and romidepsin (Istodax), were studied in relatively large phase II studies, with response rates of 25% to 30%. In a minority of patients who respond, there is some durability of response if you keep patients on therapy—sometimes a year or more—but really not a sense that this is a curative approach. A handful of other drugs (gemcitabine, bendamustine [Treanda], lenalidomide [Revlimid]) have been looked at in smaller phase II studies and have shown some activity.
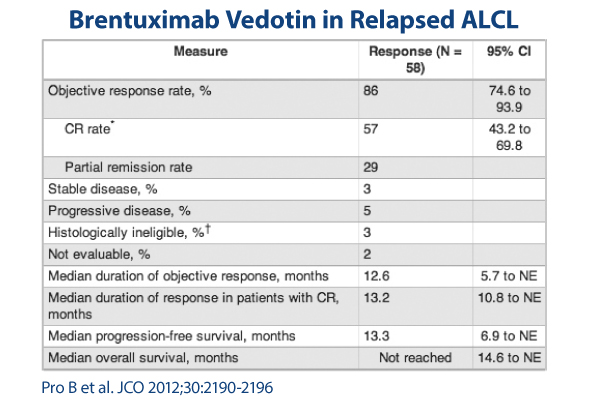
The novel agent brentuximab vedotin (Adcetris) targets CD30. Hodgkin lymphoma and anaplastic large cell lymphoma are probably the strongest, most uniform CD30-expressing lymphomas, and it looks like brentuximab vedotin hits that target very well. At least the phase II data we have in anaplastic large cell lymphoma are quite impressive (Fig. 2).
The study shown in Fig. 2 led to the approval of brentuximab for relapsed anaplastic large cell lymphoma. The overall response rate was 86%, more than half of patients with relapsed disease had complete responses, and durability was on average about a year.
The patient in this case received brentuximab vedotin, and her siblings had HLA typing performed. Neither sibling matched, but she matched unrelated donors in the registry. She had a partial response, with reduction of lymphadenopathy but residual PET uptake over background in the right axillary nodes after three and six doses of brentuximab. This woman was relatively young, with relapsed disease under good control. She went on to receive an allogeneic stem cell transplant after her seventh dose of brentuximab. ■
Suggested Readings
1. Savage KJ: ALK-negative anaplastic large cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: Report from the International Peripheral T-Cell Lymphoma Project. Blood 111:5496-5504, 2008.
2. Schmitz N, Trumper L, Ziepert M, et al: Treatment and prognosis of mature T-cell and NK-cell lymphoma: An analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood 116:3418-3425, 2010.
3. d’Amore F, Relander T, Lauritzsen GF, et al: High-dose chemotherapy and autologuos stem cell transplantation in previously untreated peripheral T-cell lymphoma: Final analysis of a large prospective multicenter study (NLG-T-01). Blood 118(suppl 21):Abstract 331, 2011.
4. O’Connor OA, Pro B, Pinter-Brown L, et al: Pralatrexate in patients with relapsed or refractory peripheral T-cell lymphoma: Results from the pivotal PROPEL study. J Clin Oncol 29:1182-1189, 2011.
5. Coiffier B, Pro B, Prince M, et al: Results from a pivotal, open-label, phase II study of romidepsin in relapsed or refractory peripheral T-cell lymphoma after prior systemic therapy. J Clin Oncol 30:631-636, 2012.
6. Pro B, Advani R, Brice P, et al: Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large-cell lymphoma: Results of a phase II study. J Clin Oncol 30:2190-2196, 2012.
7. Le Gouill S, Milpied N, Buzyn A, et al: Graft-versus-lymphoma effect for aggressive T-cell lymphomas in adults: A study by the Société Francaise de Greffe de Moëlle et de Thérapie Cellulaire. J Clin Oncol 26:2264-2271, 2008.