
Over the past several decades, progress in the treatment of acute myeloid leukemia (AML) in the older population (generally considered to be older than 60 years) has been limited (Fig. 1). In particular, the outcome of patients over age 70 has been poor, with few long-term survivors. Although AML is predominantly a disease of older adults, until recently many of the trials had been conducted in the younger population, and much of the available data was inapplicable to the elderly. This was largely attributable to reluctance (by both patients and physicians) to expose older patients to the toxic effects of “standard” chemotherapy. Furthermore, the few trials conducted specifically in the older population generally excluded patients with poor performance status and comorbid medical conditions, again limiting their relevance to community practice.
However, several studies have clearly demonstrated the benefit of treatment, showing an improved response rate and survival when older patients with AML receive leukemia-specific t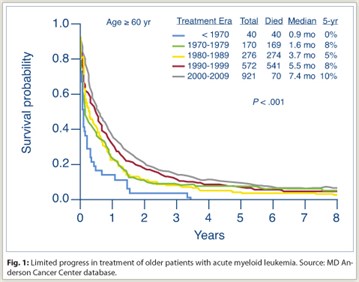 herapy, and this is not at the expense of a worsened quality of life.1,2 Using data from the Swedish Leukemia Registry, investigators have reported that the outcome of patients with AML was better in regions where they were more likely to be offered intensive induction chemotherapy.3
herapy, and this is not at the expense of a worsened quality of life.1,2 Using data from the Swedish Leukemia Registry, investigators have reported that the outcome of patients with AML was better in regions where they were more likely to be offered intensive induction chemotherapy.3
In another study, U.S. investigators using data from Medicare administrative claims linked to 11 Surveillance, Epidemiology, and End Results (SEER) registries showed that only about 30% of patients older than 65 were treated with chemotherapy, and the median survival of the entire population was only 2 months.1 Among patients over 70, fewer than 20% received chemotherapy. In a separate analysis, they compared the outcomes of matched patients treated or not treated with chemotherapy and reported approximately a three times higher median and 1-year survival in treated patients.
Patient Selection
Several studies have shown that conventional induction chemotherapy using the traditional cytarabine-plus-anthracycline regimens is only beneficial in a subset of patients in this population. Although response rates as high as 50% and 60% have been reported in such trials, the inclusion criteria for participation in these studies are generally very stringent, limiting the applicability of conclusions to only the healthiest and fittest patients, typically in the 60- to 70-year-old group.
A number of reports have examined the feasibility of intensive cytarabine-and-anthracycline–based strategies in the older AML population and have identified risk factors that predict a low response rate, a high induction mortality, and a low likelihood of 1-year survival.4 Among them, age remains an important predictor, with the outcome worse in patients over 70 and 80 years of age. Using such risk scores, it may be possible to better select patients for receiving conventional induction chemotherapy (typically those younger than 65).
 Clearly, the rest of this population should be offered investigational, less intensive strategies in a clinical trial setting. With increased understanding of the biologic mechanisms of neoplastic transformation and with the identification of the molecular events that lead to leukemogenesis, several new drugs with potential activity in AML have been developed. In many cases, these drugs are targeted at molecular events occurring in specific subgroups of patients, allowing the possibility of a personalized approach with specific drugs used for patients whose leukemia expresses the relevant target. With further advances in the field, more and more such agents will be available, and their use, either as single agents or in combination with other similar drugs, may provide us with more effective, less toxic strategies for treating the older, less fit population.
Clearly, the rest of this population should be offered investigational, less intensive strategies in a clinical trial setting. With increased understanding of the biologic mechanisms of neoplastic transformation and with the identification of the molecular events that lead to leukemogenesis, several new drugs with potential activity in AML have been developed. In many cases, these drugs are targeted at molecular events occurring in specific subgroups of patients, allowing the possibility of a personalized approach with specific drugs used for patients whose leukemia expresses the relevant target. With further advances in the field, more and more such agents will be available, and their use, either as single agents or in combination with other similar drugs, may provide us with more effective, less toxic strategies for treating the older, less fit population.
Risk-Benefit Expectations
Therefore, a decision regarding the best strategy should be based on expected outcomes. Perhaps those with the expectation of a low 8-week mortality, high complete response rate, and reasonable 3-year survival should be treated using conventional regimens, whereas those with a high expectation of initial mortality and low long-term survival should be offered investigational agents. Arguments may be offered both for and against intensive chemotherapy for patients with risk-benefit expectations between these two groups.
Clearly, conventional cytarabine/anthracycline–based strategies are inadequate in the majority of older patients with AML, particularly in those over 70. For these patients, participation in clinical studies of new agents with novel mechanisms of action is highly recommended. ■
Disclosure: Dr. Ravandi has received research funding from Sunesis, Bayer/Onyx, and Celgene. He has received honoraria from and served on the advisory board for Genzyme, Eisai, and Cephalon.
References
1. Menzin J, Lang K, Earle CC, et al: The outcomes and costs of acute myeloid leukemia among the elderly. Arch Intern Med 162:1597-1603, 2002.
2. Juliusson G, Billstrom R, Gruber A, et al: Attitude towards remission induction for elderly patients with acute myeloid leukemia influences survival. Leukemia 20:42-47, 2006.
3. Juliusson G, Antunovic P, Derolf A, et al: Age and acute myeloid leukemia: Real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood 113:4179-4187, 2009.
4. Kantarjian H, O’Brien S, Cortes J, et al: Results of intensive chemotherapy in 998 patients age 65 years or older with acute myeloid leukemia or high-risk myelodysplastic syndrome: predictive prognostic models for outcome. Cancer 106:1090-1098, 2006.
Dr. Ravandi is Associate Professor of Medicine, Department of Leukemia, University of Texas MD Anderson Cancer Center, Houston.

 Despite Advances, Little Overall Improvement Seen in Treatment of Older Adults with AML
Despite Advances, Little Overall Improvement Seen in Treatment of Older Adults with AML