The European Society for Medical Oncology (ESMO) 2018 Congress featured an assortment of study findings, many with far-reaching clinical implications for the treatment of patients with various cancers. Many of these trials were covered in-depth in recent issues of The ASCO Post. Here, we present additional highlights of studies in breast cancer to augment our ESMO coverage.
Shorter Course of Trastuzumab
For women with small HER2-positive early breast cancer, a 9-week course of trastuzumab (Herceptin) may be as effective as and less cardiotoxic than the standard 1-year course, according to a subgroup analysis of the ShortHER trial.1

Pierfranco Conte, MD
The ShortHER trial randomly assigned 1,254 patients with HER2-positive early breast cancer to receive either 9 weeks or 1 year of trastuzumab (plus chemotherapy in both). After a median of 6 years of follow-up, the short course did not achieve noninferiority but was associated with a reduction in the rate of cardiac toxicity, reported Pierfranco Conte, MD, of the University of Padua and Istituto Oncologico Veneto, Padua, Italy.
“The study was underpowered because of difficulties in recruiting patients in a reasonable time, so noninferiority could not be claimed based on the results,” he said. “Based on our data, 1 year of trastuzumab remains the standard treatment for women with HER2-positive early breast cancer.”
In a subgroup analysis, however, pathologic tumor size and nodal status were independent prognostic factors for disease-free survival, and three prognostic groups were identified:
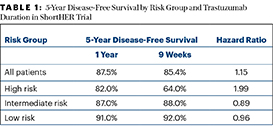
- Low risk (tumor size < 2 cm and no positive lymph nodes; 37.5% of patients)
- Intermediate risk (tumor size < 2 cm and 1 to 3 positive lymph nodes or tumor size > 2 cm and 0 to 3 positive lymph nodes; 47.1% of patients)
- High risk (any T and 4+ positive lymph nodes; 15.3% of patients).
Although high-risk patients clearly needed 1 year of trastuzumab, patients at low and intermediate risk—altogether 85% of the study population—had similar 5-year disease-free survival after a 9-week course of trastuzumab (Table 1)—with a risk of cardiac events that was nearly three times lower (4.3% vs 13.1%; relative risk = 2.8).
“Physicians can stop trastuzumab before 1 year in patients who develop a cardiac event during treatment without compromising efficacy,” Dr. Conte said. “They can consider shorter-duration trastuzumab treatment in patients at risk of cardiac toxicity and who are at low or intermediate risk of breast cancer relapse.”
Ancillary Commentary on Duration of Trastuzumab
Nadia Harbeck, MD, Head of the Breast Center at the University of Munich, commented on the ShortHER trial findings. “The results of this analysis showed that patients with a high tumor load definitely derive substantial

Nadia Harbeck, MD

Veronique Dieras, MD
benefit from a longer duration of trastuzumab…. Although it is an exploratory analysis of a negative trial and thus does not meet the criteria to be practice-changing, I think the results will influence clinicians and patients; for patients who cannot complete 1 year of trastuzumab, those with a low tumor burden may be reassured that they have not lost out on efficacy.”
Veronique Dieras, MD, of the Centre Eugène Marquis, Rennes, France, also noted that in the similarly designed SOLD trial, 9 weeks of trastuzumab was not noninferior to 1 year of treatment.2 She indicated a meta-analysis of the SOLD and ShortHER trials is underway. Meanwhile, she concluded, “One year of trastuzumab is still the standard.”
Preventing Some Cases of Cardiotoxicity
A prospective, randomized trial of 468 patients (mean age, 51 years) with HER2-positive early breast cancer found that both lisinopril, an inhibitor of angiotensin-converting enzyme (ACE), and carvedilol, a beta-blocker, significantly reduced the rate of cardiac events in patients receiving trastuzumab plus an anthracycline.3 There was no treatment effect in patients who did not receive an anthracycline.

Pamela Munster, MD
Cardiotoxicity was observed in 47% of the placebo arm, 37% of the lisinopril arm, and 31% of the carvedilol arm. In the non-anthracycline cohort, the rates of cardiotoxicity were similar: 32% with placebo, 30% with lisinopril, and 29% with carvedilol. The cardiotoxicity-free interval was also longer with lisinopril (hazard ratio [HR] = 0.53; P = .015) and carvedilol (HR = 0.49; P = .009), compared with placebo, reported Pamela Munster, MD, of the Helen Diller Family Comprehensive Cancer Center, San Francisco.
Furthermore, in patients receiving concurrent treatment with trastuzumab, fewer patients receiving the cardioprotective agents required interruptions in trastuzumab dosing. Such dose interruptions were noted in 40.3% of the placebo arm, 23.0% of the lisinopril arm, and 19.7% of the carvedilol arm (P = .007 for active treatment vs placebo), she added.
Of note, side effects were more frequent with the ACE inhibitor, especially fatigue, dizziness, headache, cough, and hypotension.
Additional Comments on Cardioprotection
Invited discussant Dr. Dieras pointed out that the population was fairly young (median age, 51 years) with good cardiac function at baseline; therefore, these patients are not necessarily representative of the typical woman with breast cancer. Documenting a protective effect in more elderly patients “would be important for translating the findings to real life,” she said, as would information on how the interventions impacted quality of life, which was not evaluated.
Since the majority of the benefit was seen in patients receiving anthracyclines, she concluded that ACE inhibitors or beta-blockers could be considered in this subgroup but then asked whether “primary prevention is the best option,” as it is “still a matter of debate.” She continued: “We need to better characterize the population at risk for heart failure, by using clinical factors, functional imaging, biomarkers, and even genetic studies (polymorphisms in the HER2 gene have been associated with risk).”
The other cardioprotective option is simply to treat patients without anthracyclines or shorten the duration of trastuzumab, which are considered acceptable for those at low risk of relapse and/or those with underlying cardiac dysfunction. “In any case,” Dr. Dieras concluded, “patients need early referral to cardio--oncologists for optimization.”
Combination Therapy for Advanced Disease
A phase III trial has found that a histone deacetylase (HDAC) inhibitor plus exemestane improves progression-free survival in patients with hormone receptor–positive/HER2-negative advanced breast cancer whose disease has progressed on prior endocrine therapy.4 The results of the ACE trial were reported at an ESMO Presidential Session by Zefei Jiang, MD, of the 307th Hospital of Chinese People’s Liberation Army in Beijing.

Zefei Jiang, MD
The oral HDAC inhibitor in the study was chidamide, which is approved for T-cell lymphoma in China but not in the United States. Several other drugs in the class are approved in the United States, and the HDAC inhibitor entinostat received Breakthrough Therapy designation by the U.S. Food and Drug Administration after the phase II ENCORE 301 trial showed a benefit (in combination with exemestane) in advanced breast cancer. Dr. Jiang added that HDAC inhibition may be able to “resensitize” tumors to estrogen modulator therapy.
In the ACE trial, 362 patients whose disease progressed on endocrine therapy were treated with either chidamide at 30 mg twice weekly plus exemestane at 25 mg daily (n = 241) or exemestane plus placebo (n = 121). The addition of chidamide nearly doubled the median progression-free survival, from 3.8 months to 7.4 months, with a hazard ratio of 0.75 (P = .0336). Objective response and clinical benefit rates also were increased. The overall survival data were immature, Dr. Jiang reported.
The most common grade 3 or higher adverse events were hematologic, including neutropenia (50.8%), thrombocytopenia (27.5%), and leukopenia (18.8%). These side effects occurred in about 2.5% of the placebo arm.

HDAC inhibitors plus endocrine therapy clearly warrants further investigation in earlier settings of hormone receptor–positive advanced breast cancer.— Rebecca Dent, MD
Tweet this quote
Invited discussant Rebecca Dent, MD, Head of Medical Oncology and senior consultant at the National Cancer Center in Singapore, noted that many patients had dose reductions or interruptions of chidamide, but otherwise the regimen showed meaningful results and “appears to be quite well tolerated.” She noted this is the second trial to show a benefit with HDAC inhibitors added to endocrine therapy, and this “clearly warrants further investigation in earlier settings of hormone receptor–positive advanced breast cancer.” She expects subgroups to be identified who may benefit most and said phase III results for entinostat are “anxiously awaited.”
Breast Cancer Vaccine in Triple-Negative Subgroup
The HER2 peptide vaccine nelipepimut-S (NeuVax), given with trastuzumab, yielded a highly significant benefit in the subset of patients with triple-negative breast cancer, in a phase IIb study at the prespecified interim analysis, reported Diane F. Hale, MD, of the San Antonio Military Medical Center in Texas.5

Diane F. Hale, MD
Nelipepimut-S plus granulocyte-macrophage colony-stimulating factor (GM-CSF) has proved to be a safe and immunogenic combination with some clinical efficacy. Preclinical studies and translational data strongly suggest there could be synergy between the vaccine and trastuzumab. This phase IIb vaccine study investigated whether a combination of trastuzumab and nelipepimut-S could prevent disease recurrence in patients with HER2 low-expressing tumors (1–2+ by immunohistochemistry).
The study’s 275 patients with HER2 low-expressing tumors were considered at high risk for recurrence (ie, were node-positive or node-negative and hormone receptor–negative [triple-negative]). Patients who were disease-free after standard therapy were randomly assigned to receive trastuzumab and nelipepimut-S (vaccine group) or trastuzumab plus GM-CSF (control group). Nelipepimut-S or GM-CSF was given every 3 weeks for 6 doses, starting with the third trastuzumab dose, and then boosted every 6 months for 4 more doses. All patients received 1 year of trastuzumab.
ESMO 2018 CONGRESS: BREAST CANCER TREATMENTS IN BRIEF
- A 9-week course of trastuzumab may be acceptable in patients with small tumors and a low risk for recurrence, according to a subanalysis of the ShortHER trial.
- In patients with early breast cancer receiving trastuzumab and an anthracycline, both lisinopril and carvedilol significantly reduced the rate of cardiac events.
- The HDAC inhibitor chidamide plus exemestane improved progression-free survival in patients with hormone receptor–positive/HER2-negative advanced breast cancer who had disease progression on prior endocrine therapy, in a study from China.
- The nelipepimut-S vaccine, given with trastuzumab, significantly improved recurrence-free survival in a subset of patients with triple-negative breast cancer, in a phase IIb study that was otherwise considered to be negative.
After a median follow-up of 19.6 months for the interim analysis, recurrence in the intent-to-treat population was observed in 13.7% of the control arm and 9.6% of the vaccine arm (P = .257), with a 2-year disease-free survival rate of 82.5% vs 88.6% (HR = 0.67, confidence interval [CI] = 0.33–1.35). “The final analysis, with a median follow-up of 25.7 months, showed a trend toward a difference in median disease-free survival favoring the combination arm (HR = 0.62; P = .175),” Dr. Hale reported.
In contrast, in the triple-negative subgroup of the interim analysis, there were recurrences in 7.5% of the vaccine group vs 26.7% of the control group (P = .023). The 2-year disease-free survival rate was 69.9% with placebo and 91.1% with nelipepimut-S plus trastuzumab (HR = 0.26, CI = 0.09–0.9; P = .023). This was again confirmed in the final analysis at 26.1 months of median follow-up (HR = 0.26, CI = 0.08–0.81; P = .013). “We saw a clinically meaningful and statistically significant difference in median disease-free survival in favor of the combination arm,” she said.
“The prespecified interim analysis shows efficacy trends overall in favor of the [nelipepimut-S] plus trastuzumab combination but most importantly demonstrates a highly significant clinical benefit in triple-negative breast cancer,” she said. “We believe a future confirmatory phase III study in this underserved population with high risk of recurrence and death is warranted.” ■
DISCLOSURE: The ACE trial was funded by Chipscreen Biosciences, and the nelipepimut-S study was funded by Sellas Life Sciences Group. Dr. Conte is on the speakers bureau of Novartis, Roche, Eli Lilly, and AstraZeneca; and has received grants/research support from Roche, Novartis, and Merck Serono. Dr. Harbeck has received honoraria for lectures and consulting from Agendia, Amgen, AstraZeneca, Celgene, Daiichi-Sankyo, Genomic Health, Lilly, MSD, NanoString, Novartis, Odonate, Pfizer, Roche, Sandoz/Hexal, and Seattle Genetics. Dr. Dieras has received honoraria from Roche/Genentech, Novartis, Lilly, Pfizer, Eisai, Nektar, Astellas, AbbVie, MSD, Tesaro, Daiichi Sankyo, Odonate Therapeutics, and Seattle Genetics. Drs. Munster and Jiang reported no conflicts of interest. Dr. Dent has received honoraria from AstraZeneca, Roche/Genentech, Pfizer, and Lilly; has been a consultant/advisor for Roche, Pfizer, Merck, Eisai, and AstraZeneca; and has received reimbursement for travel/accommodations/expenses from Roche, Pfizer, and Amgen. Dr. Hale reported no conflicts of interest.
REFERENCES
1. Conte P, Guarneri V, Bisagni G, et al: 9 weeks versus 1 year adjuvant trastuzumab for HER2+ early breast cancer: Subgroup analysis of the ShortHER trial allows to identify patients for whom a shorter trastuzumab administration may have a favorable risk/benefit ratio. 2018 ESMO Congress. Abstract 191PD_PR. Presented October 20, 2018.
2. Joensuu H, Fraser J, Wildiers H, et al: Effect of adjuvant trastuzumab for a duration of 9 weeks vs 1 year with concomitant chemotherapy for early human epidermal growth factor receptor 2-positive breast cancer: The SOLD randomized clinical trial. JAMA Oncol 4:1199-1206, 2018.
3. Guglin M, Krischer J, Tamura R, et al: Randomized trial of lisinopril or carvedilol for the prevention of cardiotoxicity in patients with early stage HER2-positive breast cancer receiving trastuzumab. 2018 ESMO Congress. Abstract 192PD. Presented October 20, 2018.
4. Jiang Z, Li W, Hu X, et al: Phase III trial of chidamide, a subtype-selective histone deacetylase inhibitor, in combination with exemestane in patients with hormone receptor-positive advanced breast cancer. 2018 ESMO Congress. Abstract 283O_PR. Presented October 20, 2018.
5. Hale DF, Clifton GT, Vreeland TJ, et al: Prespecified interim analysis of a randomized phase 2b trial of trastuzumab plus nelipepimut-S (NeuVax) vs trastuzumab for the prevention of recurrence demonstrates benefit in triple negative (HER2 low-expressing) breast cancer patients. 2018 ESMO Congress. Abstract 1128O. Presented October 22, 2018.

