Can aggressive treatment of high-risk smoldering multiple myeloma patients prevent disease worsening? A carfilzomib (Kyprolis)-based regimen and autologous stem cell transplant (ASCT) plus maintenance produced encouraging outcomes in the phase II GEM-CESAR study by the Spanish Myeloma Group. The results were reported at the 2017 American Society of Hematology (ASH) Annual Meeting & Exposition by Maria-Victoria Mateos, MD, of the University Hospital of Salamanca in Spain.1
Among patients diagnosed with smoldering myeloma, there are different subgroups with different risks of disease progression to myeloma. The phase III QuiRedex study, also conducted by the Spanish Myeloma Group, showed that treatment with lenalidomide (Revlimid) plus dexamethasone reduced the risk of disease progression in high-risk patients.2 At 75 months’ median follow-up, the median time to disease progression was not reached for the treatment arm and was 23 months with observation alone (hazard ratio = 0.24; P < .0001). Progression to multiple myeloma occurred in 39% and 86% of the arms, respectively.
GEM-CESAR Details
Subsequently, the same researchers conducted the phase II single-arm GEM-CESAR trial, in which they evaluated what they called a “curative approach” using carfilzomib-based regimens for induction and consolidation, followed by maintenance therapy. The study enrolled 126 patients at high risk (≥ 50%) of disease progression according to the Mayo, Spanish, or both risk models. Patients with a biomarker predictive of imminent risk of progression to myeloma were included, but if bone disease was detected on imaging during screening, these patients were excluded.

This curative approach for high-risk smoldering multiple myeloma seems to be encouraging. Depth of response improved over the treatment; 90% of patients receiving maintenance are in complete remission, with a 60% rate of minimal residual disease negativity.— Maria-Victoria Mateos, MD
Tweet this quote
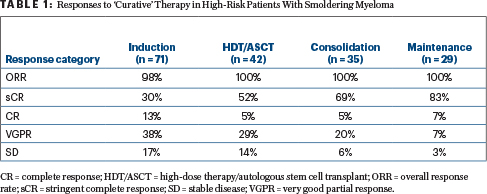
There were 36 “screening failures,” primarily due to lytic lesions but also to comorbidities, end-organ damage, withdrawal of informed consent, and false high-risk determination. This resulted in a population of 90 patients, of whom 71 received six 4-week cycles of induction therapy with carfilzomib at 36 mg/m2 twice weekly, lenalidomide at 25 mg on days 1 to 21, and dexamethasone weekly at 40 mg (KRd); 42 patients received high-dose therapy with melphalan at 200 mg/m2 followed by ASCT; 35 patients received two cycles of consolidation KRd with carfilzomib plus lenalidomide at 25 mg days 1 to 21, and dexamethasone at 40 mg a week; 29 went on maintenance therapy with lenalidomide at 10 mg on days 1 to 21 and dexamethasone at 20 mg weekly for up to 2 years.
The primary endpoint was achievement of minimal residual disease negativity after induction, high-dose therapy/ASCT, consolidation, maintenance and 3 and 5 years postmaintenance. Minimal residual disease was evaluated by next-generation flow cytometry after induction, ASCT, consolidation, and annually thereafter.
Treatment Outcomes
After a median follow-up of 10 months, 94% of patients were progression-free and 98% were alive at 28 months, Dr. Mateos reported. Response rates following each step in the treatment process are shown in Table 1. Minimal residual disease negativity increased as patients progressed through treatment, ultimately reaching 62% after consolidation (with similar rates for high-risk and ultra–high-risk patients); complete responses or better were achieved by 74% of patients, she noted.
EARLY TREATMENT OF SMOLDERING MYELOMA
- The Spanish Myeloma Group identified 90 high-risk patients with smoldering myeloma and treated them with carfilzomib-based induction, autologous transplant, carfilzomib-based consolidation, and lenalidomide maintenance.
- At 28 months, 94% of patients were progression-free and 98% were alive.
- Minimal residual disease negativity increased as patients progressed through treatment, ultimately reaching 62% after consolidation; complete responses or better were achieved by 74% of patients.
Treatment-related adverse events were rare. No patients discontinued consolidation, but two developed grade 3/4 neutropenia during this time, as did two during maintenance. Neutropenia, infections, and skin rash were the primary causes for dose reductions with lenalidomide and dexamethasone. No dose reductions were required for carfilzomib and melphalan.
“The safety profile was acceptable,” Dr. Mateos commented. “Although infections were the most frequent treatment-related adverse events, most were mild and manageable.”
At 28 months, progression-free survival was 94% and overall survival was 98%. Two patients experienced a relapse before completing therapy, and they proceeded to subsequent treatment. There were two deaths: one patient relapsed after a complete response, became refractory to treatment, and died, and another died due to ischemic stroke during induction.
“This curative approach for high-risk smoldering multiple myeloma seems to be encouraging,” Dr. Mateos concluded. “Depth of response improved over the treatment; 90% of patients receiving maintenance are in complete remission, with a 60% rate of minimal residual disease negativity.”
The incorporation of new imaging assessments allowed the researchers to identify screening failures in 18% because of the presence of bone disease, she added. ■
DISCLOSURE: Dr. Mateos has served as a consultant or advisor to and received honoraria from Celgene, Takeda, Amgen, and Janssen.
REFERENCES
1. Mateos MV, Lopez JM, Rodriguez-Otero P, et al: Curative strategy for high-risk smoldering myeloma (GEM-CESAR): Carfilzomib, lenalidomide and dexamethasone (KRd) as induction followed by HDT-ASCT, consolidation with KRd and maintenance with Rd. 2017 ASH Annual Meeting & Exposition. Abstract 402. Presented December 10, 2017.
2. Mateos MV, Hernández MT, Giraldo P, et al: Lenalidomide plus dexamethasone versus observation in patients with high-risk smouldering multiple myeloma (QuiRedex): Long-term follow-up of randomised, controlled, phase 3 trial. Lancet Oncol 17:1127-1136, 2016.


