In postmenopausal women without prior breast cancer, estrogen alone reduced the risk of breast cancer, not only during treatment, but for years after estrogen was stopped. It also reduced deaths as a result of breast cancer and deaths after breast cancer from all causes. However, in contrast, estrogen plus progestin was associated with an increased breast cancer incidence in the updated analysis of the Women’s Health Initiative hormone therapy trials.1

Rowan T. Chlebowski, MD, PhD
“The use of estrogen alone and the use of estrogen plus progestin have opposite effects on breast cancer,” said Rowan T. Chlebowski, MD, PhD, Investigator at the Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center. Now with 19.2 years of follow-up, postmenopausal women randomly assigned to estrogen alone had a 23% reduction in breast cancers, whereas estrogen plus progestin increased the incidence of breast cancer by 29%, he reported.
“Women were exposed to estrogen/progestin for 5.6 years, but the risk continued up to 20 years,” according to Dr. Chlebowski. “The risk doesn’t appear to be leveling off, so one can speculate it may be a lifetime risk for short-term use.”
With estrogen alone, deaths from breast cancer were also reduced by 44%, and deaths after breast cancer from all causes were reduced by 25%. In contrast, these endpoints were worsened with estrogen/progestin, based on these study findings.
Steven E. Vogl, MD, of the Bronx, New York, commented on these results after the presentation: “This is really powerful—a means of breast cancer prevention that also improves all-cause mortality.” In response, Dr. Chlebowski noted, “This is the only breast cancer intervention of any kind that has reported a reduction in deaths as a result of breast cancer.”
Discordant Findings
Dr. Chlebowski described how the influence of hormone therapy on breast cancer has remained controversial over the years. Discordant findings have been reported from observational studies and randomized trials, including two large observational studies published in 2019.
KEY POINTS
- In a 19-year follow-up of the randomized Women’s Health Initiative, which involved more than 10,000 postmenopausal women, the use of estrogen alone significantly reduced the incidence of breast cancer by 23%, whereas estrogen combined with progestin significantly increased the risk by 29%.
- Similar contrasting effects were observed for breast cancer–specific and all-cause mortality.
- These results stand in contrast to findings from observational studies.
The findings of the Women’s Health Initiative directly contradict those published this year by the Collaborative Group on Hormonal Factors in Breast Cancer and the Million Women Study. The Collaborative Group study was a meta-analysis of 58 observational studies in which women used hormone therapy for 7 to 10 years—estrogen alone, as well as estrogen plus progestin—both were associated with a significantly increased risk of breast cancer.2 In the Million Women Study, both types of hormone therapy were associated with significantly increased breast cancer mortality.3
“There have been a number of attempts to try to reconcile these findings,” noted Dr. Chlebowski. “The issue is how to interpret 58 observational findings compared with a 10,800-patient randomized controlled trial with 20 years of follow-up.”
Women’s Health Initiative: Two Randomized Trials
The Women’s Health Initiative enrolled women from 1993 to 1998 into two randomized trials. Postmenopausal women were randomly assigned to receive conjugated equine estrogens plus medroxyprogesterone acetate vs placebo (n = 16,608 with an intact uterus), which they took for a median of 5.6 years, or estrogen alone vs placebo (n = 10,739 with prior hysterectomy), which they remained on for a median of 7.2 years. All participants were followed for a median of 18.3 years and 16.1 years, respectively.
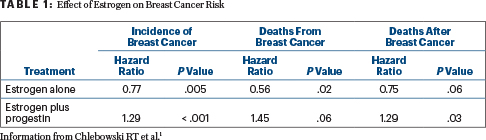
In the updated analysis presented in San Antonio, for the estrogen-alone comparison, there were 231 breast cancers in women receiving estrogen, compared with 289 for those receiving a placebo (hazard ratio [HR] = 0.77; P = .005). Women randomly assigned to estrogen alone also had a reduced risk of deaths due to breast cancer (HR = 0.56; P = .02) and for deaths after breast cancer (HR = 0.75; P = .06). By subgroup, the incidence was reduced by 55% among patients with estrogen receptor–positive/progesterone-negative disease.
In contrast, in the estrogen/progestin trial, there were 572 breast cancers diagnosed among women assigned to the combination, compared with 431 among those assigned to a placebo (HR = 1.29; P < .0001). The patients who received the combination therapy also had an increased risk for disease-specific death (HR = 1.45; P = .06) and deaths after breast cancer (HR = 1.29; P = .03; Table 1). Although it is not clear how progestin might be causing the negative effects, Dr. Chlebowksi added, “It’s well known in preclinical studies that progestins increase breast cancer stem cells in a complex process.”
No differences in outcomes were seen by age alone. “We have previously reported, for other endpoints, that women aged 50 to 59 had a much lower cardiovascular risk and improved overall survival when receiving estrogen alone, based on 17 years of follow-up,” he noted.
The reduced risk for incidence associated with estrogen alone continued beyond the termination of the 5.6-year intervention, even for decades (HR = 0.83; 95% confidence interval [CI] = 0.57–1.20). Similarly, the increased risk continued with estrogen/progestin (HR = 1.30; 95% CI = 0.99–1.70). “The sustained effects are remarkable,” commented Dr. Chlebowski. “No one predicted that. It’s a major finding of the Women’s Health Initiative.”
In closing, Dr. Chlebowski said that women who are considering estrogen alone should know that it’s safer, and there may be a breast cancer benefit associated with its use. “Women considering estrogen plus progestin have a little bit more difficult dilemma because they have to be willing to accept the maybe 20-year, maybe lifetime, increased breast cancer risk…. The first thing, however, is to go over the numbers with them. The absolute risk is still small.”
DISCLOSURE: Dr. Chlebowski has received honoraria from Novartis, AstraZeneca, and Genentech and has received consulting fees from Novartis, AstraZeneca, Pfizer, Puma, Immunomedics and Genentech. Dr. Vogl’s spouse has stock or other ownership interests in Amgen and Bristol-Myers Squibb.
REFERENCES
1. Chlebowski RT, Anderson GL, Aragaki AK, et al: Long-term influence of estrogen plus progestin and estrogen alone use on breast cancer incidence: The Women’s Health Initiative randomized trial. 2019 San Antonio Breast Cancer Symposium. Abstract GS5-00. Presented December 13, 2019.
2. Collaborative Group on Hormonal Factors in Breast Cancer: Type and timing of menopausal hormone therapy and breast cancer risk: Individual participant meta-analysis of the worldwide epidemiological evidence. Lancet 394:1159-1168, 2019.


