In patients with stage III melanoma, a reduced-dose neoadjuvant immunotherapy combination was well tolerated and led to high pathologic response rates, in the phase II OpACIN-neo trial presented at the European Society for Medical Oncology (ESMO) 2018 Congress.1
“Neoadjuvant ipilimumab (Yervoy) at 1 mg/kg plus nivolumab (Opdivo) at 3 mg/kg is less toxic than the standard dosing regimen. The high response rate was preserved, making this schedule an attractive candidate to be tested against adjuvant PD-1 [programmed cell death protein 1] blockade in a phase III study,” said Christian U. Blank, MD, PhD, of the Netherlands Cancer Institute, Amsterdam.

Neoadjuvant ipilimumab at 1 mg/kg plus nivolumab at 3 mg/kg is less toxic than the standard dosing regimen.— Christian U. Blank, MD, PhD
Tweet this quote
Adjuvant immunotherapy is now a well-established treatment of high-risk stage III melanoma, with data from large trials demonstrating an improvement in 5-year recurrence-free and overall survival with adjuvant ipilimumab and improved recurrence-free survival with adjuvant nivolumab and pembrolizumab (Keytruda). The stage is now set to move immunotherapy into the neoadjuvant setting.
“We hypothesized that neoadjuvant T-cell checkpoint blockade can induce a stronger and broader T-cell response than adjuvant immunotherapy,” Dr. Blank explained.
Previous Trials
The previous phase Ib OpACIN study evaluated standard-dose ipilimumab plus nivolumab in both the adjuvant and neoadjuvant settings.2 Neoadjuvant treatment led to greater expansion of tumor-resident T-cell–receptor clones than adjuvant treatment, and this was accompanied by a high pathologic complete response rate, 45%, after just 6 weeks of treatment.
“All these patients remain relapse-free today, after about 3 years of follow-up, but this came with a 90% rate of grade 3 to 4 immune-related toxicities, making this scheme unfeasible,” Dr. Blank said.
“Two other trials addressed whether the use of PD-1 monotherapy blockade, not a combination, was better, but the rate of pathologic complete responses was low, and some patients’ condition deteriorated, and they could not undergo surgery. We learned that PD-1 monotherapy blockade is not the way to go in the neoadjuvant setting,” he said.
OpACIN-neo Trial
The investigators then designed the multicenter phase II -OpACIN-neo trial of a reduced-dose regimen (lower doses and/or fewer courses) to determine whether toxicity could be reduced without compromising efficacy. The OpACIN-neo trial randomly assigned patients with resectable stage III melanoma and at least one measurable lymph node metastasis to one of the following regimens:
- Arm A: 2 courses of ipilimumab at 3 mg/kg + nivolumab at 1 mg/kg every 3 weeks
- Arm B: 2 courses of ipilimumab at 1 mg/kg + nivolumab at 3 mg/kg every 3 weeks
- Arm C: two courses of ipilimumab at 3 mg/kg every 3 weeks followed immediately by 2 courses of nivolumab at 3 mg/kg every 2 weeks (closed early due to toxicity).
Complete lymph node dissection was scheduled at week 6. Primary endpoints were grade ≥ 3 immune-related adverse events within the first 12 weeks, radiologic response rate (Response Evaluation Criteria in Solid Tumors, version 1.1), and pathologic response rate (< 50% viable tumor cells).
Toxicity Reduced
After a median follow-up of 8.3 months, 86 patients were evaluated. On the advice of the data safety and monitoring board, arm C was closed early due to toxicity. Although 50% of arm C experienced grade ≥ 3 immune-related adverse events, these side effects occurred in 40% of arm A and just 20% of arm B. Aside from immune-related adverse events, most other toxicities, primarily wound infections, were related to surgery.
We learned that PD-1 monotherapy blockade is not the way to go in the neoadjuvant setting.— Christian U. Blank, MD, PhD
Tweet this quote
“Reduction from 4 to 2 courses reduced toxicity from 90% in the previous OpACIN study to 40% in OpACIN-neo (arm A),” Dr. Blank noted. “In arm B, toxicity was reduced another way, to 20%, by decreasing the dose of ipilimumab. The sequential application of these drugs did not improve toxicity.”
High Radiologic and Pathologic Response Rates
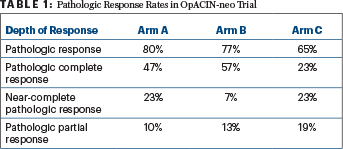
“The radiologic response was high, after only 6 weeks of treatment,” he reported. Radiologic responses were observed in 60% of arm A, 60% of arm B, and 42% of arm C, with complete radiologic responses achieved in 7%, 10%, and 4%, respectively. Pathologic responses were also high in arm A (80%) and arm B (77%) but not arm C (65%; Table 1), according to Dr. Blank.
“Radiologic responses—either partial responses or stable disease—underestimated the pathologic responses,” he said. “There were many pathologic complete responses in these groups.”
“The response rates in arm A were identical to those in the previous trial, with an 80% pathologic response rate and a 47% rate of pathologic complete response, while in arm B 77% pathologic responses with 57% rate of pathologic complete response were observed,” he added, showing that less-toxic regimens can preserve the high response rates achieved with these drugs. Event-free survival at this time point has not differed among the arms, stabilizing at approximately 80%, as in the previous trial. Two patients have died, both in arm A—one due to progression of melanoma and another due to complications of immune-related encephalitis.
NEOADJUVANT THERAPY IN MELANOMA
- Established as effective in the adjuvant treatment of stage III melanoma, neoadjuvant immunotherapy is now being evaluated in clinical trials.
- The phase II OpACIN-neo trial evaluated three different dosing schedules of ipilimumab plus nivolumab.
- Two courses of neoadjuvant ipilimumab at 1 mg/kg plus nivolumab at 3 mg/kg proved to be less toxic than standard dosing and yielded a radiologic response rate of 60% and a pathologic response rate of 77%.
- Follow-up is too short to know whether pathologic response correlates with improved survival.
MORE FROM ESMO
For more information from the 2018 ESMO Congress on research studies in advanced melanoma treatment as well as toxicities of immunotherapy in this setting, see two interviews with Caroline Robert, MD, PhD, on The ASCO Post Newsreels at www.ascopost.com/videos.
“Pathologic response is a good indicator of outcome for relapse-free survival,” he said. “Patients who had a pathologic response don’t tend to relapse, whereas patients without a pathologic response relapse early. In this study, 43% of nonresponders have relapsed so far. Also, patients with a ‘cold’ immune signature (low levels of interferon-gamma) have relapsed, whereas those with ‘hot’ signatures have not.”
OpACIN-neo confirms the high pathologic response rate observed previously in the OpACIN trial, when patients were treated with neoadjuvant ipilimumab plus nivolumab, but they were achieved at lower doses. “Two courses of ipilimumab at 1 mg/kg and nivolumab at 3 mg/kg has been identified as the safest combination and “should be the new standard,” Dr. Blank maintained. ■
DISCLOSURE: Dr. Blank has served as an advisor to Bristol-Myers Squibb, MSD, Roche, Novartis, Glaxo-Smith Kline, Pfizer, Eli Lilly, GenMab, and Pierre Fabré.
REFERENCES
1. Rozeman EA, Menzies AM, Van de Wiel BA, et al: OpACIN-neo: A multicenter phase 2 study to identify the optimal neo-adjuvant combination scheme of ipilimumab and nivolumab. 2018 ESMO Congress. Abstract LBA42. Presented October 22, 2018.
2. Blank CU, Rozeman EA, Fanchi LF, et al: Neoadjuvant versus adjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma. Nat Med 24:1655-1661, 2018.


