In the Clinic provides overviews of novel oncology agents, addressing indications, mechanisms, administration recommendations, safety profiles, and other essential information needed for the appropriate clinical use of these drugs.
Indications
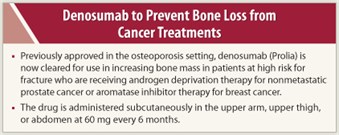 In September 2011, the monoclonal antibody RANKL inhibitor denosumab (Prolia) was approved for use in increasing bone mass in patients at high risk for fracture who are receiving androgen deprivation therapy for nonmetastatic prostate cancer or aromatase inhibitor therapy for breast cancer.1 Under the trade name Xgeva, denosumab has an indication for prevention of skeletal-related events in patients with bone metastases from solid tumors at a different dose (120 mg subcutaneously every 4 weeks) than in the current indications. Denosumab (as Prolia) also has a nononcologic indication for treatment of postmenopausal women with osteoporosis at high risk for fracture.2
In September 2011, the monoclonal antibody RANKL inhibitor denosumab (Prolia) was approved for use in increasing bone mass in patients at high risk for fracture who are receiving androgen deprivation therapy for nonmetastatic prostate cancer or aromatase inhibitor therapy for breast cancer.1 Under the trade name Xgeva, denosumab has an indication for prevention of skeletal-related events in patients with bone metastases from solid tumors at a different dose (120 mg subcutaneously every 4 weeks) than in the current indications. Denosumab (as Prolia) also has a nononcologic indication for treatment of postmenopausal women with osteoporosis at high risk for fracture.2
Approval of denosumab in the nonmetastatic breast cancer setting was based on a 2-year trial comparing every-6-month subcutaneous injections (total of four doses) of denosumab (n = 127) vs placebo (n = 125) in 252 women (median age 59 years) receiving aromatase inhibitor therapy (62% for > 6 months).3 Patients had to have bone mineral density (BMD) T-scores of –1.0 to –2.5 at the lumbar spine, total hip, or femoral neck and could not have had a fracture after age 25. Changes in lumbar spine BMD at 12 months, the primary endpoint of the trial, were  +4.8% with denosumab and –0.7% with placebo, a treatment difference of 5.5% (P < .0001).
+4.8% with denosumab and –0.7% with placebo, a treatment difference of 5.5% (P < .0001).
Approval in the nonmetastatic prostate cancer setting was based on a 3-year trial comparing every-6-month subcutaneous injections (total of six doses) of denosumab (n = 734) vs placebo (n = 734) in 1,468 men (median age 76 years) receiving androgen deprivation therapy (79% > 6 months).4 Men younger than age 70 had to have a BMD T-score between –1.0 and –4.0 at the lumbar spine, total hip, or femoral neck or history of osteoporotic fracture. Changes in lumbar spine BMD T-scores at 24 months, the primary endpoint of the trial, were +5.6% with denosumab and –1.0% with placebo, a treatment difference of 6.7% (P < .0001). Denosumab treatment also resulted in a 66% relative risk reduction (1.5% vs 3.9%, P = .0125) for new vertebral fracture at 36 months, a secondary endpoint of the trial.
How It Works
 Both androgen deprivation therapy and aromatase inhibitor therapy are associated with bone loss and risk of fracture. Denosumab is a monoclonal antibody that binds to RANKL, a protein involved in the formation, function, and survival of osteoclasts, the cells responsible for bone resorption.5 By inhibiting resorption, denosumab inhibits bone loss.
Both androgen deprivation therapy and aromatase inhibitor therapy are associated with bone loss and risk of fracture. Denosumab is a monoclonal antibody that binds to RANKL, a protein involved in the formation, function, and survival of osteoclasts, the cells responsible for bone resorption.5 By inhibiting resorption, denosumab inhibits bone loss.
How It Is Given
Denosumab should be administered by a health-care professional. The recommended dose is 60 mg every 6 months as a subcutaneous injection in the upper arm, upper thigh, or abdomen. Patients should take 1,000 mg of calcium and at least 400 IU of vitamin D daily. Hypocalcemia must be corrected prior to starting denosumab; it may worsen, particularly in patients with renal impairment. Patients receiving denosumab as Xgeva should not receive denosumab as Prolia (same active ingredient). The dose of denosumab (Prolia) in the prostate and breast cancer settings is the same as that for postmenopausal women with osteoporosis.
Safety Profile
 In the trials supporting approval of denosumab, adverse events reported in at least 10% of denosumab patients and seen more frequently than in placebo patients were arthralgia and back pain. Pain in extremities and musculoskeletal pain were also observed more frequently with denosumab treatment. In men receiving androgen deprivation therapy, denosumab was associated with a greater frequency of cataracts (4.7% vs 1.2%). Hypocalcemia was observed only in denosumab patients (2.4%) at 1 month after the first dose. Warnings and precautions for denosumab include hypocalcemia, serious infections including skin infections, osteonecrosis of the jaw, and oversuppression of bone turnover.
In the trials supporting approval of denosumab, adverse events reported in at least 10% of denosumab patients and seen more frequently than in placebo patients were arthralgia and back pain. Pain in extremities and musculoskeletal pain were also observed more frequently with denosumab treatment. In men receiving androgen deprivation therapy, denosumab was associated with a greater frequency of cataracts (4.7% vs 1.2%). Hypocalcemia was observed only in denosumab patients (2.4%) at 1 month after the first dose. Warnings and precautions for denosumab include hypocalcemia, serious infections including skin infections, osteonecrosis of the jaw, and oversuppression of bone turnover.
Cost
The drug cost for denosumab is approximately $825 per 60-mg injection, or $1,650 for two injections per year. ■
References
1. U.S. Food and Drug Administration: What’s New from the Office of Hematology Oncology Products: Denosumab (Prolia). Available at http://www.fda.gov/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/ucm272420.htm. Accessed December 9, 2011.
2. PROLIA® (denosumab) injection for subcutaneous use prescribing information. Amgen Inc, September 2011. Available at http://pi.amgen.com/united_states/prolia/prolia_pi.pdf. Accessed December 9, 2011.
3. Ellis GK, Bone HG, Chlebowski R, et al: Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for nonmetastatic breast cancer. J Clin Oncol 26:4875-4882, 2008.
4. Smith MR, Egerdie B, Hernández Toriz N, et al: Denosumab in men receiving androgen-deprivation therapy for prostate cancer. N Engl J Med 361:745-755, 2009.
5. Muir VJ, Scott LJ: Denosumab: In cancer treatment-induced bone loss. BioDrugs 24:379-386, 2010.

